The demand for batteries is expected to increase 10x by 2030 due to climate change driving the move to renewable energy and electric vehicles^. The GWh demand from EVs alone is forecast to grow more than five times by 2025, and more than 50 times by 2040.
Li‑ion batteries can’t be made significantly smaller or lighter and fast charging times and cycle life are also reaching their limits.
This means there needs to be a breakthrough in battery technology to drive clean energy innovation and adoption.
Put simply — the world needs a better battery.
Lithium sulfur is superior to lithium ion in many ways.
*Comparative life cycle assessment of Li-Sulphur and Li-ion batteries for electric vehicles report. Benveniste et al, 2022.
Lithium metal batteries
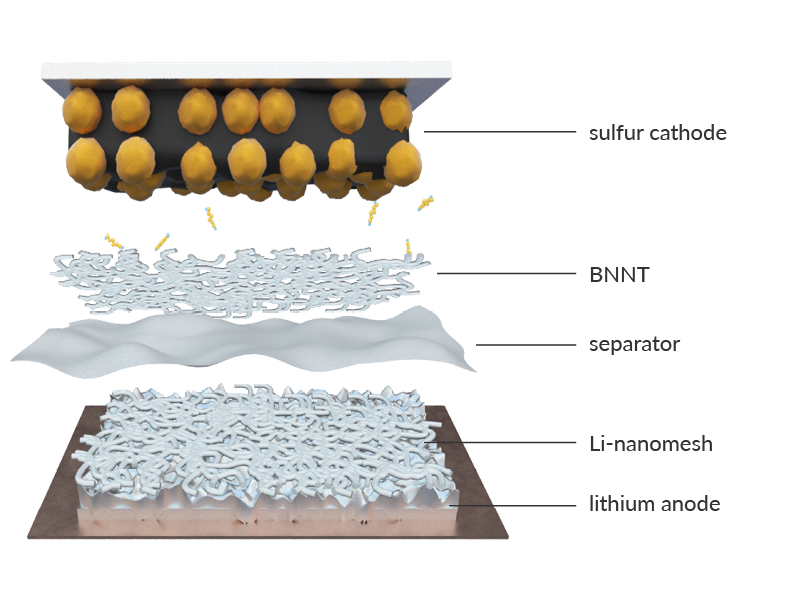
In addition to our breakthrough lithium sulfur technology, our scientists are able to use the same Li-nanomesh™ nanocomposite to enhance a second revolutionary battery: lithium metal.
This battery type has a unique advantage in that while it doesn’t quite match the energy density of lithium sulfur, it can accommodate higher power draws — a quality favoured by electric cars, scooters and multi-prop drones.
BNNT — the game-changing nano material
Boron Nitride Nanotubes (BNNTs) are an advanced nanomaterial with unique physical properties. NASA has identified it as a key nanomaterial that may even help humans reach Mars, due to its strength and radiation absorption properties. We take advantage of its physical strength, thermal conductivity and electrical resistance as well as some unique electrochemical properties to enhance our lithium sulfur and lithium metal batteries
Historically, BNNTs have been very difficult to make in high purities and at high volume — and were expensive — so were disregarded as a solution for many real-world problems, including battery electrochemistry.
However, in 2020 Australian company BNNTTL successfully commissioned a high volume BNNT production facility, commercialising Deakin University’s patented BNNT manufacturing technology and know-how.
Now, through our partnership with BNNTTL, Li-S Energy not only has all the BNNT required to commercialise Li-S technology, but also exclusive distribution rights to sell BNNTs to other battery manufacturers.